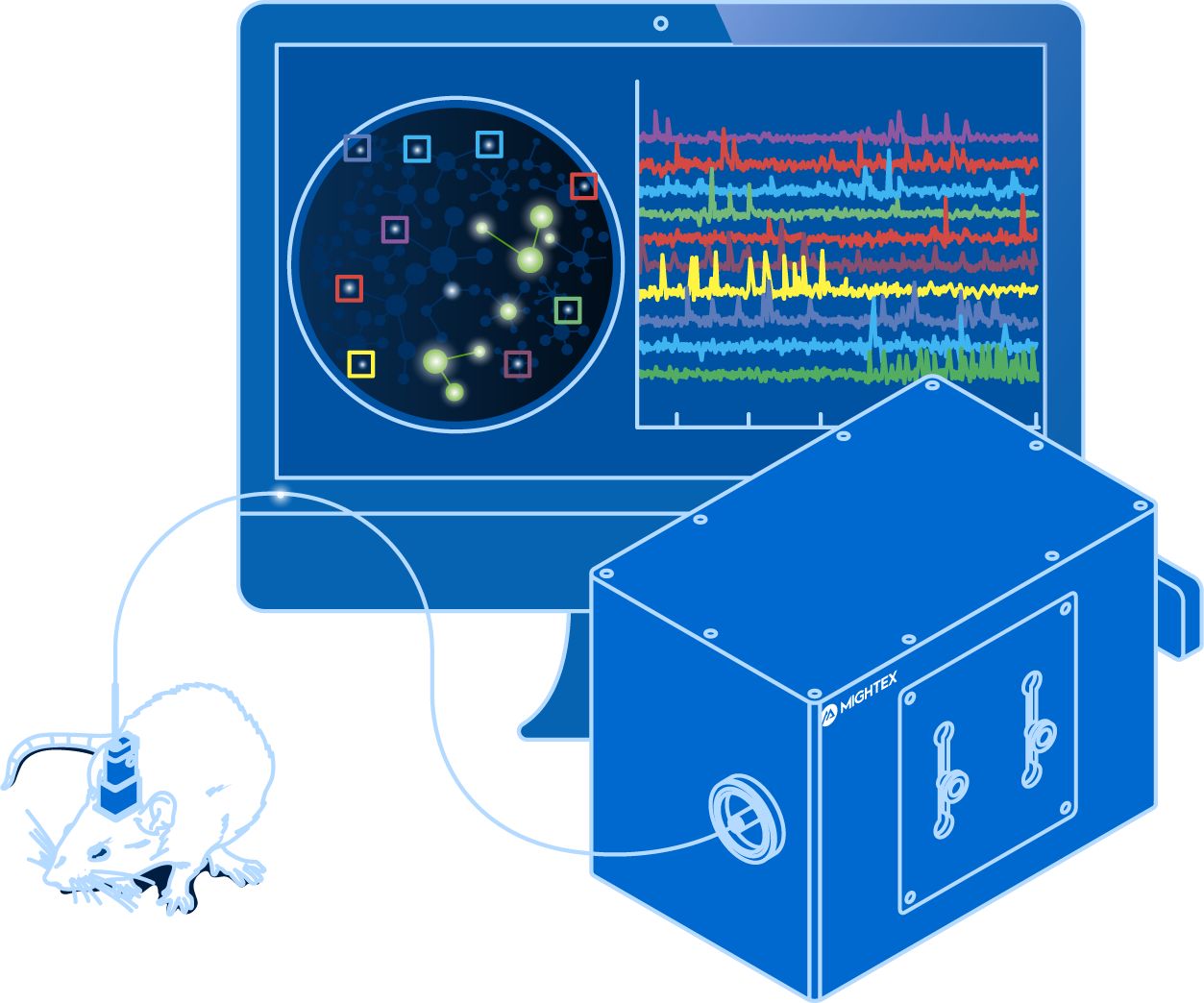
Electrophysiology is the golden standard for recording individual neuron activity. By recording electrical changes across the cell membrane, electrophysiology is able to detect action potentials in real-time (Petersen, 2017). Due to the complex nature of the technique, however, electrophysiology cannot be used to measure the activity of hundreds of neurons simultaneously, although such capability is essential for understanding the complex interactions between populations of neurons leading to cognition, behaviour, and sensory processing.
Calcium imaging has helped scientists overcome this limitation by enabling them to visualize the activity of large neuron populations. Using genetically-encoded calcium indicators, scientists are able to measure neural activity changes in hundreds of neurons simultaneously through fluctuations in fluorescence signals (Grienberger & Konnerth, 2012). Although calcium imaging has enabled the ability to visualize large neural population activity, it relies upon an indirect indicator of neural activity (i.e.calcium influx), which is inherently very slow, and hence, it is unable to faithfully reflect the relatively fast neuron activity (Grienberger & Konnerth, 2012). Electrophysiology, on the other hand, is much faster and it is able to reflect real-time changes in neural activity.
Is there a technique that can combine the strengths of both electrophysiology and calcium imaging to measure neural activity?
What is Voltage Imaging?
Building upon the strengths of electrophysiology and calcium imaging, voltage imaging enables scientists to visualize neural activity using fluorescent activity sensors that are sensitive to changes in voltage (Knopfel & Song, 2019). Unlike calcium imaging that relies on an indirect indicator, voltage imaging reflects real-time neuronal firing, and thus, it is fast enough to detect action potentials and subthreshold responses (Knopfel & Song, 2019). At the same time, voltage Imaging enables scientists to visualize a large number of neurons simultaneously.
The development of genetically encoded voltage indicators (GEVIs) has enabled neuroscientists to study specific cell-types (e.g. excitatory or inhibitory neurons). Some of the recently developed GEVI’s include ASAP3, Voltron, QuasAR3, and SomArchon (Villette et al., 2019; Abdelfattah et al., 2019; Adam et al., 2019; Piatkevich et al., 2019). However, since the field of voltage imaging is quite new, the GEVI toolbox is constantly growing with new indicators optimized for certain experiments, such as two-photon imaging.
Why Use Voltage Imaging?
Voltage imaging, being a relatively new technique, is employed by only a few labs and currently being optimized for more global use. The potential of voltage imaging’s capabilities can help advance the ability of tracking real-time neural activity in large neuron populations.
Voltage imaging visualizes a direct indicator of activity by measuring changes in voltage, similar to electrophysiology. Measuring electrical changes, or action potentials, provides a method to neural activity changes in real-time with high speed, reflecting the key marker of neural activity (Knopfel & Song, 2019). This is in contrast to calcium imaging which measures the indirect and slow indicator of calcium influx (Grienberger & Konnerth, 2012).
GEVIs enable the visualization of specific neural populations, similar to calcium imaging. Compared to electrophysiology that can measure only a few neurons simultaneously, voltage imaging can identify and measure the activity of multiple neurons simultaneously to track activity long-term.
Voltage imaging has the ability to measure neural activity, reflecting electrical changes, in behaving animals. This provides scientists with the highest temporal and spatial precision to track neural activity over time in multiple neurons to correlate neuron firing, behaviour, and cognition. Electrophysiology can be performed in vivo; however, it cannot be used to spatial track large populations of neurons in vivo, making voltage imaging an essential technique for recording neural activity in vivo.
Due to the nature of an early-stage technique, voltage imaging has a few challenges associated with both the biology and equipment, which need to be properly addressed as the technology continues to evolve.
Voltage indicators are fast, reflecting millisecond changes in neural activity, and, as a result, voltage imaging requires a high frame-rate scientific camera (Knopfel & Song, 2019; Yang & St. Pierre, 2016). Some cameras currently available in the market are able to reach high enough frame rates, but they may require cropping the field of view to reach the adequate speed, thereby limiting the field of view to a smaller number of neurons (Knopfel & Song, 2019; Yang & St. Pierre, 2016).
In addition, a fast frame rate also leads to shorter exposure time, meaning that a camera used for voltage imaging must also have high sensitivity, in order to capture significant signals from GEVIs (Knopfel & Song, 2019; Yang & St. Pierre, 2016).
Furthermore, stronger light sources can help reach the required light intensity; however, higher light intensity on the sample increases the likelihood of photobleaching the sample, decreasing the length of the experiment (Knopfel & Song, 2019; Yang & St. Pierre, 2016). Some research labs are optimizing the brightness of GEVis to help resolve this problem. Through the development of calcium imaging, there has been a significant increase in brightness of light sources, so given time these problems should be properly addressed.
Another issue with voltage imaging is low signal-to-noise ratio. This is associated with high light scattering and relatively low expression in tissue (Knopfel & Song, 2019; Yang & St. Pierre, 2016). Scientists are also working on optimizing GEVis to improve the signal-to-noise ratio.
To summarize, while calcium imaging is currently the most well-established technique for visualizing large neural circuits in vivo, voltage imaging may become the future of imaging neural activity, thanks to its superior real-time accuracy. Further development of brighter, more sensitive, better signal-to-noise, and better expression will help make voltage imaging a powerful tool.
Next Post
What Do You Need to Perform Voltage Imaging?